The ADAMs gene family of cellular sheddases are exciting, new therapeutic targets and biomarkers emerging within several clinical arenas.
ADAM is an acronym for “A Disintegrin And Metalloproteinase” which highlights two key protein modules present in the structural organisation of this family of single-span, integral membrane proteins (Fig. 2). In humans, twenty five family members have been identified of which half are thought to be catalytically active due to the presence of the consensus zinc-binding HEXXH motif within the metalloproteinase domain. ADAMs belong to the Adamalysin subgroup of the metzincin superfamily of metalloproteinases which also include the secreted ADAMs with thrombospondin motifs (ADAMs-TS) and the snake venom metalloproteinases (SVMPs) (1-3). As with MMPs, ADAMs are translated as inactive zymogens with an inhibitory prodomain masking access to the metalloproteinase domain catalytic cleft. Following activation, ADAMs regulate key proteolytic events such as the cleavage of membrane-bound ligands and receptors within their ectodomains (shedding), thereby regulating cell signalling. ADAMs also participate in the regulated intramembranous proteolysis (RIPing) of membrane proteins facilitating the release of their intracellular domains often for translocation to the nucleus (2, 3). ADAMs can also regulate cell-cell adhesion either positively through the action of their disintegrin domains which bind to cognate integrin receptors on adjacent cells or negatively through the shedding of cadherin ectodomains.
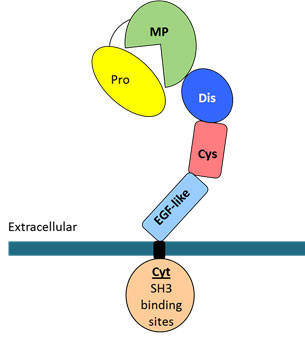 Fig. 2 Schematic representation of an ADAM sheddase. ADAMs have a modular structure with an N-terminal metalloprotease domain (MP) regulated by a pro-domain (Pro), a disintegrin domain (Dis), cysteine-rich (Cys), and EGF-like domain, a single membrane spanning helix and a C-terminal cytoplasmic domain harbouring several SH3 binding sites (Cyt).
Fig. 2 Schematic representation of an ADAM sheddase. ADAMs have a modular structure with an N-terminal metalloprotease domain (MP) regulated by a pro-domain (Pro), a disintegrin domain (Dis), cysteine-rich (Cys), and EGF-like domain, a single membrane spanning helix and a C-terminal cytoplasmic domain harbouring several SH3 binding sites (Cyt).
Through their cellular action and upregulation in diseased tissues, ADAMs sheddases have been implicated in the progression of several pathologies most notably the cancers, where they have been associated with regulating EGF/Her signalling, Notch activation and tumour cell invasiveness (2-4). There is increasing clinical and research data implicating ADAMs in the pathophysiology of inflammatory conditions such as rheumatoid arthritis and chronic airway diseases including asthma and COPD (5, 6 & Table 1). ADAMs have also been implicated in promoting pathological angiogenesis which is essential for tumour growth and metastasis and in the progression of ocular diseases that can lead to blindness. However, many studies, particularly those employing genetic linkage analyses, do not identify whether the genetic alteration leads to a loss or gain of ADAM function or in the case of expression analysis, whether elevated levels of ADAMs promote or limit disease progression. This underscores the need for the pharmacological validation of target ADAM therapeutic candidates and why Vasgen’s AbIMP technology, in the absence of selective chemical inhibitors of ADAMs catalytic activity, offers a new approach to fill the technological gap.
Citations:
- Gomis-Ruth, FX. (2009). Catalytic Domain Architecture of Metzincin Metalloendoproteinases. J Biol. Chem. 284, 15353-15358.
- Murphy G. (2008). The ADAMs: Signalling scissors in the tumour microenvironment. Nature Reviews Cancer. 8, 929-941.
- Blobel CP. (2005). ADAMs: Key players in EGF signalling, development and disease. Nature Revs Mol. Cell. Biol. 6, 32-45.
- D.R. Edwards, M.M. Handsley, C.J. Pennington (2008). The ADAM metalloproteinases. Mol Aspects Med 29, 258-289.
- Holgate ST. (2010). ADAM metallopeptidase domain 33 (ADAM33): identification and role in airways disease. Drug News Perspect.;23(6):381-7.
- Mocchegiani E, Giacconi R, Costarelli L. (2011). Metalloproteases/anti-metalloproteases imbalance in chronic obstructive pulmonary disease: genetic factors and treatment implications. Curr Opin Pulm Med. 17 Suppl 1:S11-9.
